Constant Potential vs OCP or vs Reference Electrode
There is sometimes confusion about the option to measure versus Ecorr / OCP and versus the reference electrode. In this section the reasons for choosing a certain DC potential is explained. The advantages of using Ecorr as a reference point will be presented as well as how to choose in PSTrace to use potentials versus the reference electrode or versus Ecorr.
Why measure versus the Open Circuit Potential (OCP)?
As mentioned before during EIS an AC voltage is applied and the current answer is measured. The AC voltage should be applied in a potential region, where a small potential change induces a big answer in current. This means, if we plot the current versus the potential, we want to choose a potential range where the slope of the reaction we are interested in is high.
A simple system to demonstrate this is a platinum electrode in a solution containing [Fe(CN)6]3- and [Fe(CN)6]4-. The non-diffusion limited voltammogram of this species shows clearly that the biggest current changes are induced around the formal potential of the species (see Figure 6.15), which is in agreement with the Nernst Equation (equation 3.4). If a DC potential is applied, which corresponds to the formal potential, the AC potential will have good sensitivity.
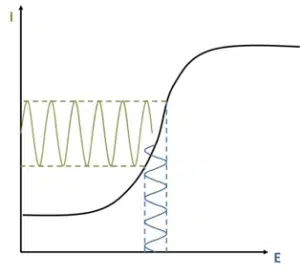
For the example of a [Fe(CN)6]3- and [Fe(CN)6]4- solution the formal potential can be looked up in a table or measured by a linear sweep voltammetry for example. This value can be entered as E dc in the corresponding field of PSTrace.
Another method is to use a 1-to-1 ratio of [Fe(CN)6]3- and [Fe(CN)6]4-. The immersed electrode’s potential (OCP) will be the formal potential of [Fe(CN)6]3- and [Fe(CN)6]4-. This has two advantages: Identification of the right potential and keeping a steady state. The right potential is the OCP. Even if an unusual reference electrode is used, the potential to be applied is quite clear. It can be easily determined by a short open circuit potential (OCP) measurement also known as corrosion potential measurement.
Furthermore, the system will not be disturbed when the measurement starts. If you move the potential away from the OCP, the environment of the electrode will react and the system won’t be in a stable state, but a diffusion layer will grow. If the measurement happens at the OCP of the electrode, this means the electrode had the potential before the measurement anyway. As a result only the AC potential will “wiggle” at the equilibrium.
For corrosion measurements it is very common to measure at the Ecorr, which corresponds to the OCP, of the sample. This potential usually represents a stable state at least in the time scale of the EIS measurement. If the sample wouldn’t change at all, no corrosion would happen and the measurement would be unnecessary in the first place. This means, if a sample is observed over a long time, the Ecorr will change. If before each EIS measurement the Ecorr is determined and used as the E dc the measurements will still be comparable and deliver sensitive measurements.
EIS versus OCP in PSTrace
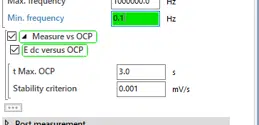
The possibility to measure the EIS versus the OCP is present in PSTrace. You just need to check the checkbox Measure vs OCP and E dc versus OCP. The first checkbox triggers an OCP measurement directly before the EIS recording is performed. The second checkbox changes the reference point for the E dc. In electronics only potential differences can be measured, so the potentiostat needs to have a 0 reference point. This is usually the reference electrode, so 0.25 V in E dc usually means 0.25 V more anodic than the reference electrode. If the check box E dc versus OCP is checked, a 0.25 V in E dc means 0.25 V more anodic than the Ecorr. The same is applicable to other techniques, for example for polarization curves.
The two additional parameters that are displayed define the duration of the OCP measurement (see Figure 6.16). The t Max. OCP is the longest time the OCP measurement takes. If the OCP measurement reaches this duration, it will be stopped and the last measured value is used as OCP. The OCP measurement can be shorter if the Stability criterion is met. If the change of the OCP per second is lower than the Stability criterion, the OCP is considered stable and the OCP measurement is stopped. The last recorded value is used as OCP.